Febraury 12, 2016
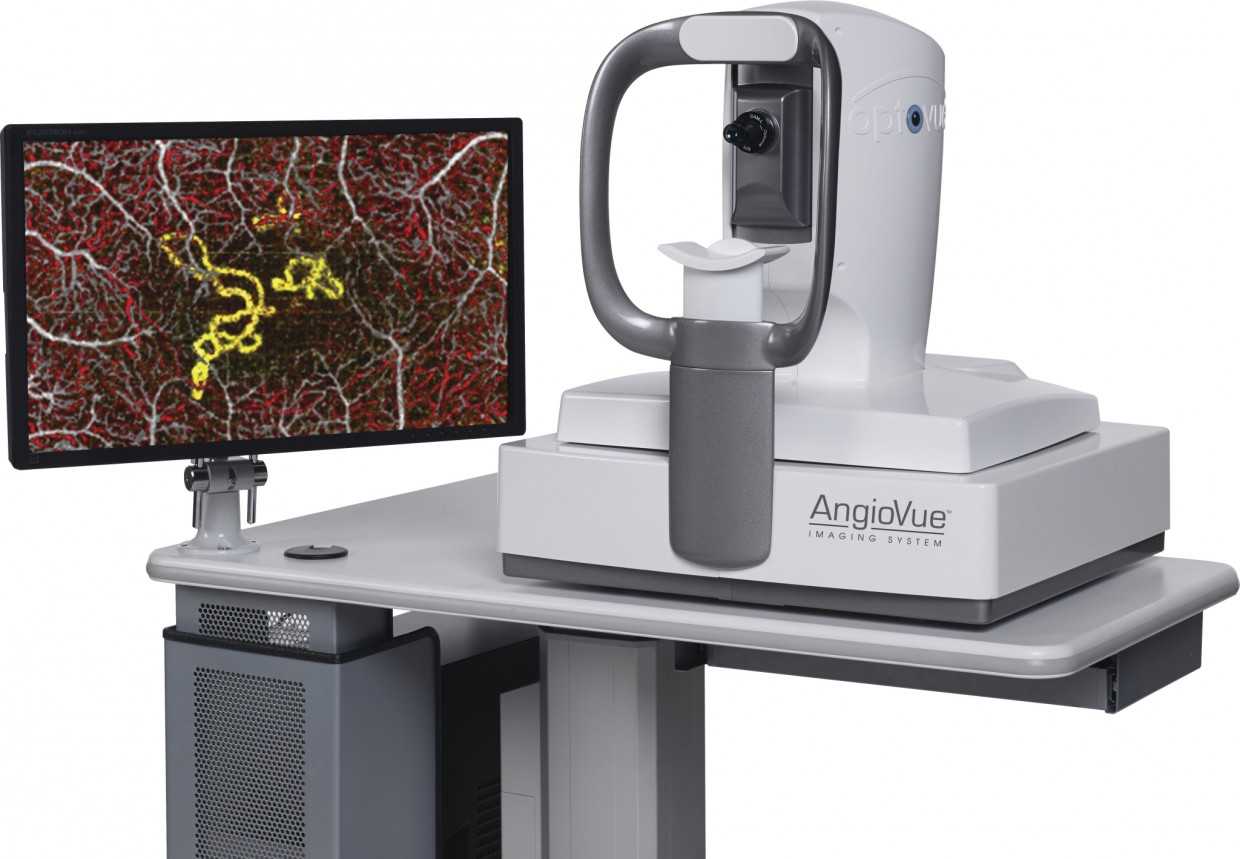
“Optovue, the global leader in the development and commercialization of optical coherence tomography angiography (OCTA), today announced FDA clearance and immediate U.S. commercial availability of the AngioVue™ Imaging System, a groundbreaking technology that provides a non-invasive way to visualize abnormal blood vessels in the retina to help physicians determine an appropriate course of treatment.”
Read more here!: http://www.optovue.com/news/optovues-angiovue-imaging-system-receives-fda-clearance/